 Society
Society

Ninety scientific reports on recent advancements in treating diseases of the liver, pancreas and other related organs were presented at the 15th national congress of the Việt Nam Association for the Study of Hepatological Diseases that closed in HCM City last Saturday (Nov 30).
|
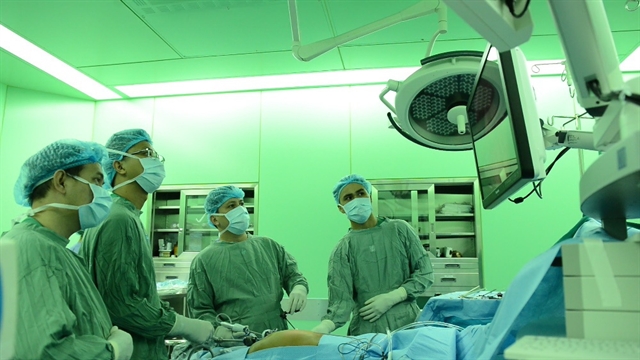
| Doctors at the University Medical Centre perform a laparoscopic liver surgery. — Photo courtesy of the hospital |
HCM CITY — Ninety scientific reports on recent advancements in treating diseases of the liver, pancreas and other related organs were presented at the 15th national congress of the Việt Nam Association for the Study of Hepatological Diseases that closed in HCM City last Saturday.
This year’s congress, which had the theme “Update on Diagnosis and Treatment of Hepatology Diseases” was attended by more than 600 local and foreign doctors and experts in the hepato-pancreato-biliary field.
Prof Lê Gia Vinh, deputy president of the association, said the congress offered the country’s doctors a significant opportunity to compare notes with their peers in the hepato-biliary-pancreatic field.
Việt Nam has consistently adopted advanced technologies for the examination and treatment of hepatological diseases, resulting in better quality of diagnosis and treatment, he said.
Hepato-biliary-pancreatic surgeons boast significant achievements in recent years, he said.
Martin Smith, president of the International Hepato-Pancreato-Biliary Association, said: “I am sure that the knowledge, sharing and experience will be very significant and valuable to all of us.
“What you have achieved in Việt Nam is really something that our chapters in many countries around the world should learn from.”

|
A doctor checks a patient with liver disease. — VNA/VNS Photo
At the two-day congress, Vietnamese and foreign experts presented the latest developments in the diagnosis and treatment of diseases such as hepatitis, cirrhosis, liver cancer, organ transplant, image diagnosis, and minimally invasive surgeries.
The association promised to strengthen dissemination of research findings and best treatment practices, foster research and innovation and encourage international collaboration. — VNS




