 Society
Society

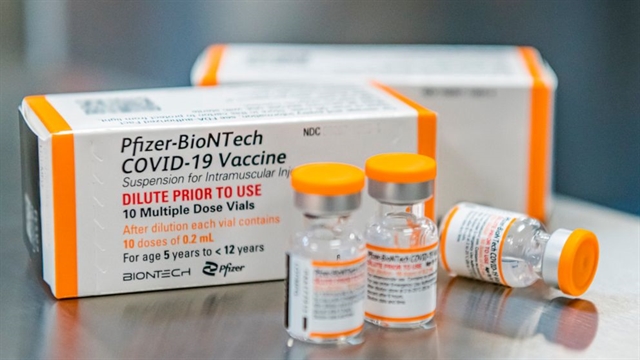
|
| Pfizer/BioNTech COVID-19 vaccines for children aged 5 to under 12 years old. — Photo from Pfizer |
HÀ NỘI — The Ministry of Health on Friday officially granted approval of Pfizer/BioNTech COVID-19 vaccines for use in children aged five to under 12 years old.
The decision was reflected in the amended approval for emergency use of the Comirnaty shots originally signed in June last year.
According to the decision, one dose for children from 12 years and above is 0.3ml of vaccine (after dilution) containing 30mcg of mRNA enveloped by lipid nanoparticles, while the one dose for children from five to under 12 is 0.2ml of vaccine (after dilution) containing a third of the amount of mRNA for adults, or 10mcg.
With regards to packaging, for people aged 12 years and older, the products are placed into trays with 195 vials in each tray, and one vial holding six doses of vaccines; or placed into a box of 25 vials, with one vial holding six doses; or placed in one box of 10 vials, with one vial also holding six doses.
The paediatric vaccines are placed in either a tray of 195 vials with each vial containing 10 doses, or a box of 10 vials with each vial holding 10 doses.
The manufacturers of the vaccines are listed as Pfizer Manufacturing Belgium NV (Belgium);, BioNTech Manufacturing GmbH (Germany), Pharmacia and Upjohn Company LLC (the United States), and Hospira Incorporated (the United States).
The decision takes effect from today.
In last week's meeting with National Assembly deputies on schooling in COVID-19, deputy health minister Đỗ Xuân Tuyên said 98 per cent of adults (aged 18 and above) in Việt Nam had received two doses of vaccines, and booster shot coverage was at 32 per cent, while 94 per cent of children aged 12-17 had been fully vaccinated (also using Pfizer shots).
The procedures to purchase 21.9 million doses of Pfizer vaccines shots to use for 10 million children in the 5-11 age group had been “basically completed,” he said.
Prime Minister Phạm Minh Chính has asked localities and the health sector to prepare all conditions, including facilities, storage and transport, and human resources to carry out the COVID-19 inoculation of children aged 5-11 years right after the vaccines are delivered, which is expected to be at the end of April this year. — VNS




