 Society
Society

More than 1,000 volunteers have received their second jab of the domestically developed COVID-19 vaccine Nano Covax in its third phase of human trials, the Defence Ministry’s Military Medical University has announced.
|
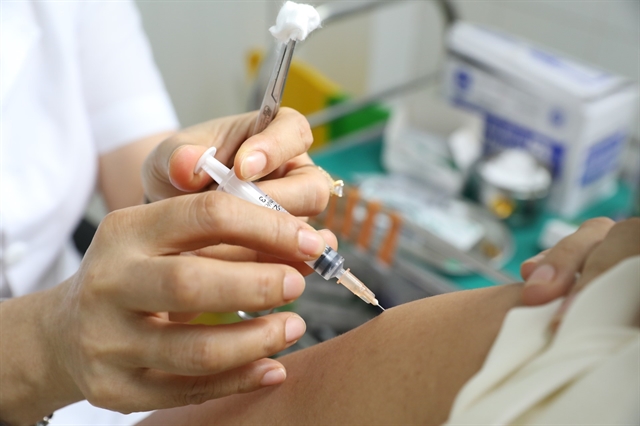
| Việt Nam's domestically developed COVID-19 vaccine Nano Covax is administered to a volunteer. — VNA/VNS Photo Minh Quyết |
HÀ NỘI — More than 1,000 volunteers have received their second jab of the domestically developed COVID-19 vaccine Nano Covax in its third phase of human trials, the Defence Ministry’s Military Medical University has announced.
Prof. Dr. Maj. Gen. Hoàng Văn Lương, Deputy Director of the university, which is in charge of administering the doses and carrying out medical evaluations on the volunteers before and after the shots, said there had been no significant adverse reactions.
Volunteers are required to update information regarding any side effects they might experience on an e-Diary.
More than 12,000 remaining volunteers will get their second dose from July 26.
All volunteers are expected to have been given two shots of Việt Nam’s homegrown vaccine by mid-August.
Volunteers in phase three are between the age of 18 and 75 and have had their blood tested for the presence of antibodies to the SARS-CoV-2 virus.
People who have been exposed to SARS-CoV-2 or already have antibodies have been excluded.
Nano Covax is developed by the Nanogen Pharmaceutical Biotechnology JSC based on recombinant DNA/protein technology. It began first-phase trials in December 2020 and second phase trials in February of this year.
Results from the first two trial phases showed that all volunteers developed antibodies against the coronavirus SARS-CoV-2, but more data on how long that protection lasts is needed.
If the results of the phase three trials are positive and Nano Covax is given emergency approval from the National Biomedicine Ethics Council, the vaccine could be ready for large scale distribution as early as November. — VNS
Watch our video on the construction of field hospitals in HCM City




