

At least 100,000 doses of the meningococcal vaccine will be imported to Việt Nam by the end of May, increasing the number of imported doses to some 800,000 in 2018.
 |
| Việt Nam will import 800,000 doses of the meningococcal vaccine this year. — vietnamplus.vn |
HÀ NỘI — At least 100,000 doses of the meningococcal vaccine will be imported to Việt Nam by the end of May, increasing the number of imported doses to some 800,000 in 2018.
According to Đỗ Văn Đông, deputy head of the Drug Administration under the Ministry of Health (MoH), the two types of meningococcal vaccine that are registered and allowed to be circulated in the domestic market are imported from foreign countries.
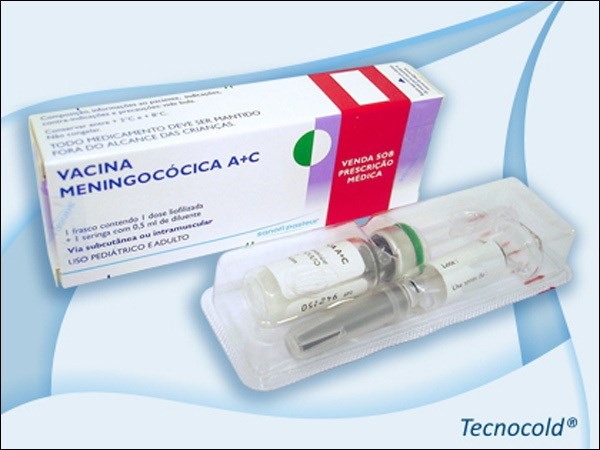
These are French-made Polysaccharide Meningococcal A+C and Cuban-made VA-Mengoc-BC. According to the representative office of Sanofi Pasteur, the manufacturer of Polysaccharide Meningococcal A+C, the company has stopped producing the vaccines worldwide because several countries are switching to meningococcal A, C, Y and W‐135 polysaccharide‐protein conjugate vaccines.
Therefore, only small quantities of Polysaccharide Meningococcal A+C and VA-Mengoc-BC vaccines are being stocked. However, meningococcal A, C, Y and W‐135 polysaccharide‐protein conjugate vaccines are yet to be licensed.
At least 125,000 doses of the VA-Mengoc-BC vaccine were imported to Việt Nam on April 20. Of this, 49,000 doses passed the test of the National Institute of Vaccines and Biologicals, while the rest are being further tested before being supplied to vaccination facilities.
To ensure adequate and timely supply of vaccines, the administration has asked provincial departments of health to direct local preventive health centres and vaccination facilities to prepare plans to purchase and stock vaccines.
Hospitals under MoH, the National Institute of Hygiene and Epidemiology, HCM City Pasteur Institute, Nha Trang City Pasteur Institute and the Central Highlands Institute of Hygiene and Epidemiology have also been asked to prepare detailed plans in estimating the demand for vaccines and stockpiling them. — VNS




.jpg)