 Society
Society

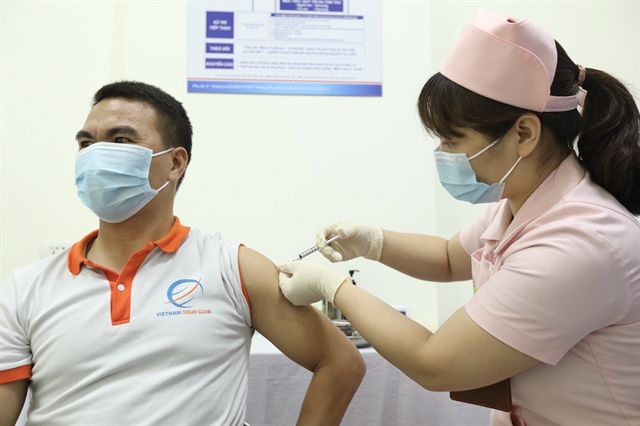
|
| The second shot of COVIVAC, Việt Nam's second COVID-19 vaccine to enter human trials, in the first phase is administered to 15 volunteers on Tuesday. — VNA/VNS Photo |
HÀ NỘI — Việt Nam expects to have the first batch of locally-produced COVID-19 vaccines ready at the end of the third quarter of 2021.
The statement was made by Deputy Director of the Department of Science Technology and Training under the Health Ministry Nguyễn Ngô Quang, who is Chief of the Office of the National Programme on Vaccine Research and Development, at a meeting of the Standing Board of the Steering Committee for International Cooperation in Clinical Trials of COVID-19 Vaccines on Monday.
Việt Nam is currently researching three types of COVID-19 vaccine.
Accordingly, the second phase of human trials for the NanoCovax vaccine, produced by the Nanogen Pharmaceutical Biotechnology, is being conducted, with the second dose to be injected on March 26.
The results of the second phase are expected to be announced in May as scheduled, while the third phase will be conducted from May to September. The vaccine is hoped to be registered for circulation in September, three months sooner than planned. Earlier, the trial time of the second phase was also shortened from six months to three months.
A system on monitoring and assessing vaccines’ protection efficiency in Việt Nam and other countries is expected to be operated from September 2021-September 2022.
Meanwhile, the first jabs of the first phase of human trials on Việt Nam’s second homegrown candidate vaccine COVIVAC, developed by the Institute of Vaccines and Medical Biologicals (IVAC), were administered on March 15. The second shot in the first phase was administered to 15 volunteers on Tuesday.
The first phase of human trials for the country’s third homegrown candidate vaccine, developed by VABIOTECH, is hoped to begin in April.
Permanent members of the Steering Committee for International Cooperation in Clinical Trials of COVID-19 Vaccines agreed that the outcomes of pre-clinical trials of all three vaccines were assessed as good thanks to Vietnamese units’ close coordination with prestigious vaccine producers and research units around the world and their compliance with international standards and procedures in vaccine research and development.
Speaking at the meeting, Deputy Prime Minister Vũ Đức Đam, who is head of the National Steering Committee for COVID-19 Prevention and Control, hailed efforts made by the Ministries of Health and Science and Technology, and vaccine research and development units.
He requested extra efforts to speed up domestically-developed vaccine research and production.
The official noted that vaccines might need repeated shots, administered every year, to deal with mutations and new variants of SARS-COV-2.
Experts and scientists said WHO is deliberating guidelines on vaccine trials conducted based on comparisons with approved vaccines, which might help facilitate Việt Nam’s phase 3 trial of locally made COVID-19 vaccine.
At the event, the health ministry affirmed that it had no policy that allows businesses and companies to import COVID-19 vaccines and administer the shots on their own.
Only the vaccines that the health ministry has approved for emergency use in Việt Nam (which to date, comprise Oxford/AstraZeneca, Moderna, and Sputnik V) can be imported by companies given authorisation to do so, and the administration of COVID-19 vaccines must follow the coordination of the health ministry, which prioritises high-risk groups first in line with the Government’s Decree.
The vaccine jabs can only be given at health ministry-approved medical establishments. — VNS




