 Society
Society

Medical equipment and machinery used in hospitals across Việt Nam require significant upgrades, and the management for these expensive devices is currently lacking.
 |
| 401-Info1 |
HÀ NỘI — Medical equipment and machinery used in hospitals across Việt Nam require significant upgrades, and the management for these expensive devices is currently lacking.
The issue received a lot of attention from National Assembly deputies during sessions held last month, especially following a shocking incident where faults in maintaining and operating kidney dialysis machines caused eight deaths in the northern province of Hoà Bình in late May.
However, the discussion mostly revolved around flawed policies and regulations that have caused significant losses for the state budget.
Recent cases drawing public attention took place at general hospitals in the Hoài Đức and Thường Tín rural districts of Hà Nội. According to documentation, the automated biochemical analysis equipment purchased by the two hospitals were imported from Germany. In fact, the machines turned out to be cheaper versions that were manufactured in China.
Recently released findings by the State Audit showed that a lot of newly bought equipment has either fallen into a state of disrepair or sustained damage, rendering them unusable even before being put into operation.
One of the most scandalous revelations was the fraudulent purchase of medical equipment in Gia Lai Province in the Central Highlands region: In the period between 2013-2015, the actual cost of medical equipment, VNĐ5.6 billion (US$246,000), was inflated to VNĐ16.7 billion ($734,600) in falsified documents.
Nguyễn Thị Kim Tiến, Minister of Health, in her answers to inquiries at the NA meeting, confirmed the veracity of reports on broken equipment, especially at provincial level public hospitals, and attributed this phenomenon to “being overused.”
However, many NA deputies were dissatisfied with the Health Minister’s answers, which they deemed ‘ambiguous.’ NA deputies hoped the government and the NA would soon arrive at appropriate measures to prevent incidents relating to medical equipment and avoid mistakes that may come at the cost of patients, as modern equipment is a necessity but decisions need to be practical and mindful of the budget situation.
Ass. Prof. Phạm Khánh Phong Lan, head of HCM City Food Safety Management Board, also a member of HCM City NA delegation, said that due to the fact that the cost of medical equipment is drawn from state budgets, hospitals would try to get as much as possible. “Hospitals may sometimes say in reports that they need state-of-the-art and expensive devices, but the number of those capable of using such devices or the number of patients actually in need of them are not enough, but if they don’t say they need it this year then next year, the budget will be cut,” she said, adding that state co-ordination must take into account these nuances to effectively manage state resources and prevent waste.
Loose management
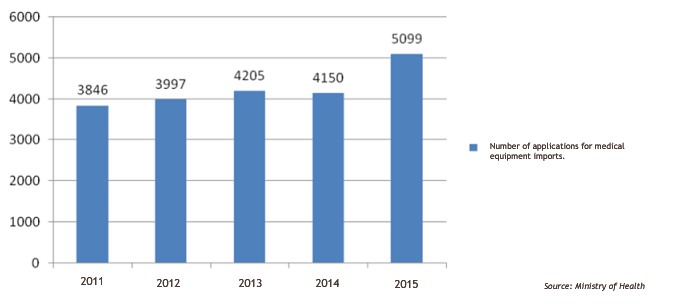
According to the Ministry of Health (MoH), in the last 5 years, the number of applications for medical equipment imports has been on the rise.
In 2011, there were 3,846 applications, two years later, the figure saw a slight increase to 4,025, but in 2015, the figure jumped to 5,099 applications.
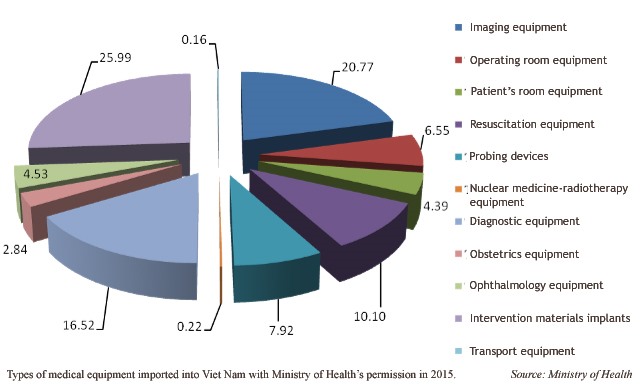
In 2015, for example, the four major groups of the applications for medical equipment were intervention materials and implant devices (26 per cent), imaging medical equipment (20.7 per cent), diagnostic devices (16 per cent), and resuscitation equipment (10 per cent).
However, the Department of Medical Equipment under the Ministry of Health said they were not able to get hold of the longevity of the medical equipment imported into the country, since “the actual figure also involves other sectors as well.”
The State Audit found that the Ministry of Health had not properly categorised medical equipment or chemicals according to types, technical standards appropriate to each medical service; and the Ministry had not established a database on bidding prices for medical centres in the country to be able to plan ahead.
Health Minister Tiến said the MoH had submitted Decree 36 on managing medical equipment and purchasing procedures to the NA Standing Committee.
The regulations provided in this legislation were claimed to be ‘relatively foolproof,’ with decentralised management taking a focus as well as more assessment of the usage of medical equipment at local levels.
In addition, MoH has developed schemes like the establishment of a Patient Safety Council at the ministry’s Department of Treatment Management as well as in hospitals. — VNS
 |
| 401-Info |




